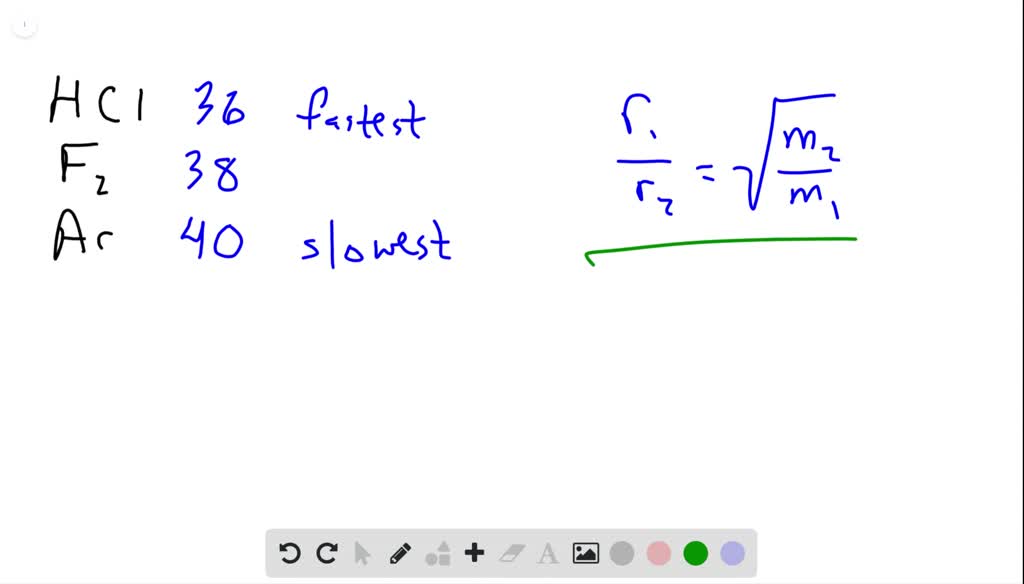
The ratio of rates of diffusion of gases X and Y is 1:5 and that of Y and Z is 1:6. The ratio of rate of diffusion of Z and X is:

Which of the following pair of gases will have same rate of diffusion under similar conditions? - YouTube

The ratio of rate of diffusion of gases A and B is `1 : 4`. If the ratio of their masses present in - YouTube

acid base - What is the relative rate of diffusion of ammonia to hydrogen chloride, both in gaseous states? - Chemistry Stack Exchange
Can the rate of effusion or diffusion be negative, in accordance with Graham's law? If so, how? - Quora

SOLVED:The rate of diffusion of a gas is proportional to: (a) (P)/(√(d)) (b) (P)/(d) (c) √((P)/(d)) (d) (√(P))/(d)

If the ratio of the rates of diffusion of two gases A and B is 4:1 the ratio of their density is - YouTube